
An isotope of uranium has an atomic number of 92 and a mass number of 235.What are the atomic number and the mass number of these carbon atoms? The most common carbon atoms have six protons and six neutrons in their nuclei.However, as we shall see later, this will not always be the case. Neutral atoms have the same number of electrons as they have protons, so their overall charge is zero. The sum of the number of protons and neutrons in the nucleus is called the mass number The sum of the number of protons and neutrons in a nucleus. A very rare form of hydrogen has one proton and two neutrons in the nucleus this isotope of hydrogen is called tritium. This particular isotope of hydrogen is called deuterium. However, a small number (about one in a million) of hydrogen atoms have a proton and a neutron in their nuclei. For example, most hydrogen atoms have a single proton in their nucleus. Most naturally occurring elements exist as isotopes. Atoms of the same element (i.e., atoms with the same number of protons) with different numbers of neutrons are called isotopes Atoms of the same element that have different numbers of neutrons. Each element has its own characteristic atomic number.Ītoms of the same element can have different numbers of neutrons, however. Thus, hydrogen has an atomic number of 1, while iron has an atomic number of 26. This number of protons is so important to the identity of an atom that it is called the atomic number The number of protons in an atom. 
All atoms of hydrogen have one and only one proton in the nucleus all atoms of iron have 26 protons in the nucleus. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons. 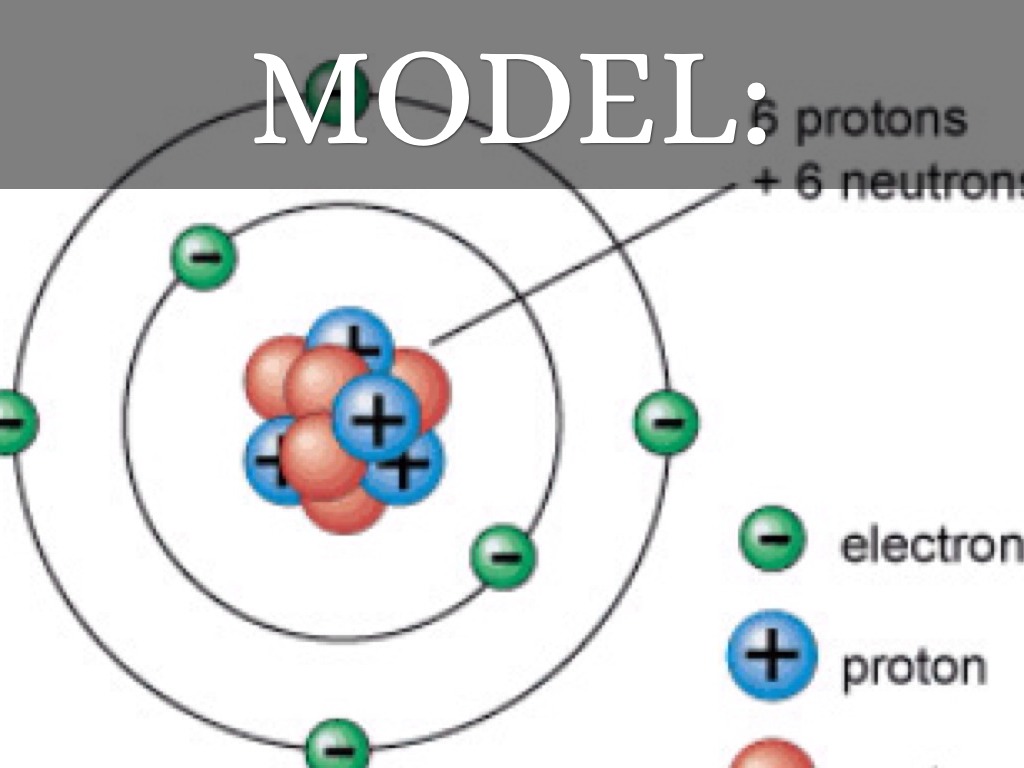
The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. Table 3.1 "Properties of the Three Subatomic Particles" summarizes the properties of these three subatomic particles.Ītoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. We now know that all atoms of all elements are composed of electrons, protons, and (with one exception) neutrons. is a subatomic particle with about the same mass as a proton but no charge. The neutron A subatomic particle with no charge. is a more massive (but still tiny) subatomic particle with a positive charge, represented as p +. The proton A subatomic particle with a positive charge. Later, two larger particles were discovered. It is often represented as e −, with the right superscript showing the negative charge. The first part to be discovered was the electron A tiny subatomic particle with a negative charge., a tiny subatomic particle with a negative charge. These concepts form the basis of chemistry.Īlthough the word atom comes from a Greek word that means “indivisible,” we understand now that atoms themselves are composed of smaller parts called subatomic particles. Atoms combine in whole-number ratios to form compounds.Atoms of the same element are the same atoms of different elements are different.The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory The concept that atoms play a fundamental role in chemistry., first stated by John Dalton, an English scientist, in 1808. Atoms are so small that it is difficult to believe that all matter is made from atoms-but it is. The period at the end of a printed sentence has several million atoms in it. It would take about fifty million atoms in a row to make a line that is 1 cm long. This 2-to-1 ratio means that the brown compound has twice the amount of chlorine per amount of copper as the green compound.The smallest piece of an element that maintains the identity of that element is called an atom The smallest piece of an element that maintains the identity of that element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
